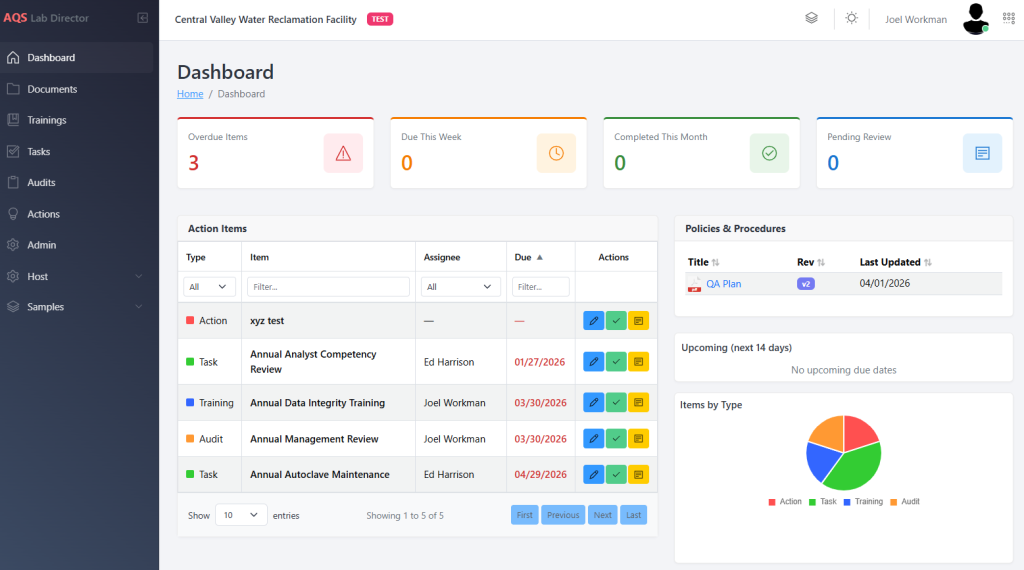
Laboratory Quality Management Software Built for Environmental Laboratories
QA Manager is a cloud-based platform designed specifically for environmental laboratories to manage quality systems, training, and ongoing compliance activities.
It brings tasks, SOPs, training, documentation, and quality records into a single, structured workflow—helping laboratories operate more consistently, efficiently, and defensibly.
Instead of relying on spreadsheets, shared drives, and manual tracking, QA Manager provides a clear, organized system for managing laboratory quality activities.
Designed for Laboratory Operations
Maintaining a laboratory quality system involves coordinating training, tracking recurring tasks, managing SOPs, documenting activities, and preparing for assessments.
QA Manager is built around how laboratories actually operate—supporting day-to-day quality activities while improving consistency, visibility, and accountability across the organization.
Why Laboratories Use QA Manager
Many laboratories rely on a combination of spreadsheets, calendars, shared folders, and paper records to manage quality activities.
Over time, this can lead to:
- missed or overdue quality tasks
- inconsistent documentation and recordkeeping
- difficulty tracking training and staff responsibilities
- limited visibility into overall quality system status
- challenges preparing for audits and assessments
QA Manager brings these activities together into a structured workflow—helping laboratories stay organized, reduce manual tracking, and maintain a more complete and defensible quality record.
Core Capabilities
Task Management & Recurring Activities
Plan, assign, and track recurring quality tasks across the laboratory.
QA Manager allows laboratories to schedule activities such as instrument checks, calibrations, training, and routine quality requirements—with due dates, assigned staff, and required documentation.
Tasks are automatically generated based on defined schedules, helping ensure that required activities are completed on time and consistently documented.
Training & Personnel Tracking
Manage employee training and competency in a structured, traceable system.
Track required training such as data integrity, method-specific requirements, MDLs, and demonstrations of capability—linked to laboratory methods, matrices, and analytes.
QA Manager provides clear visibility into training status by employee, supporting both internal management and external audits.
SOPs & Document Control
Maintain controlled standard operating procedures and supporting documents.
QA Manager supports document organization, version tracking, and consistent access to current procedures—helping ensure staff are working from the correct and approved documentation.
Quality Records & Documentation
Capture and maintain complete records of laboratory quality activities.
QA Manager provides a centralized location for attaching evidence of completed tasks, recording observations, and maintaining documentation needed to support laboratory operations and audits.
Audit Preparation & Readiness
Stay prepared for internal and external assessments.
By organizing tasks, training, and documentation within a single system, QA Manager helps laboratories demonstrate compliance more efficiently—reducing last-minute preparation and improving audit readiness.
Visibility & Oversight
Maintain clear visibility into laboratory quality system status.
QA Manager provides a structured view of upcoming tasks, completed activities, and outstanding requirements—helping laboratory managers monitor performance and identify gaps before they become issues.
Configurable to Your Laboratory
Every laboratory operates differently.
QA Manager can be configured to reflect your laboratory’s methods, matrices, analytes, personnel structure, and quality requirements—allowing you to improve organization and consistency without forcing a rigid or generic system.
Designed to Work with AB Manager
QA Manager and AB Manager are designed to support complementary roles within the accreditation process.
Laboratories use QA Manager to implement and maintain quality systems.
Accrediting bodies use AB Manager to review, verify, and coordinate those systems.
Together, they align laboratory operations with accreditation expectations—supporting more consistent, transparent, and defensible outcomes.
See QA Manager in Action
QA Manager can be configured to support your laboratory’s workflow, staff, and quality system requirements.
Contact AQS to schedule a demonstration or learn more.